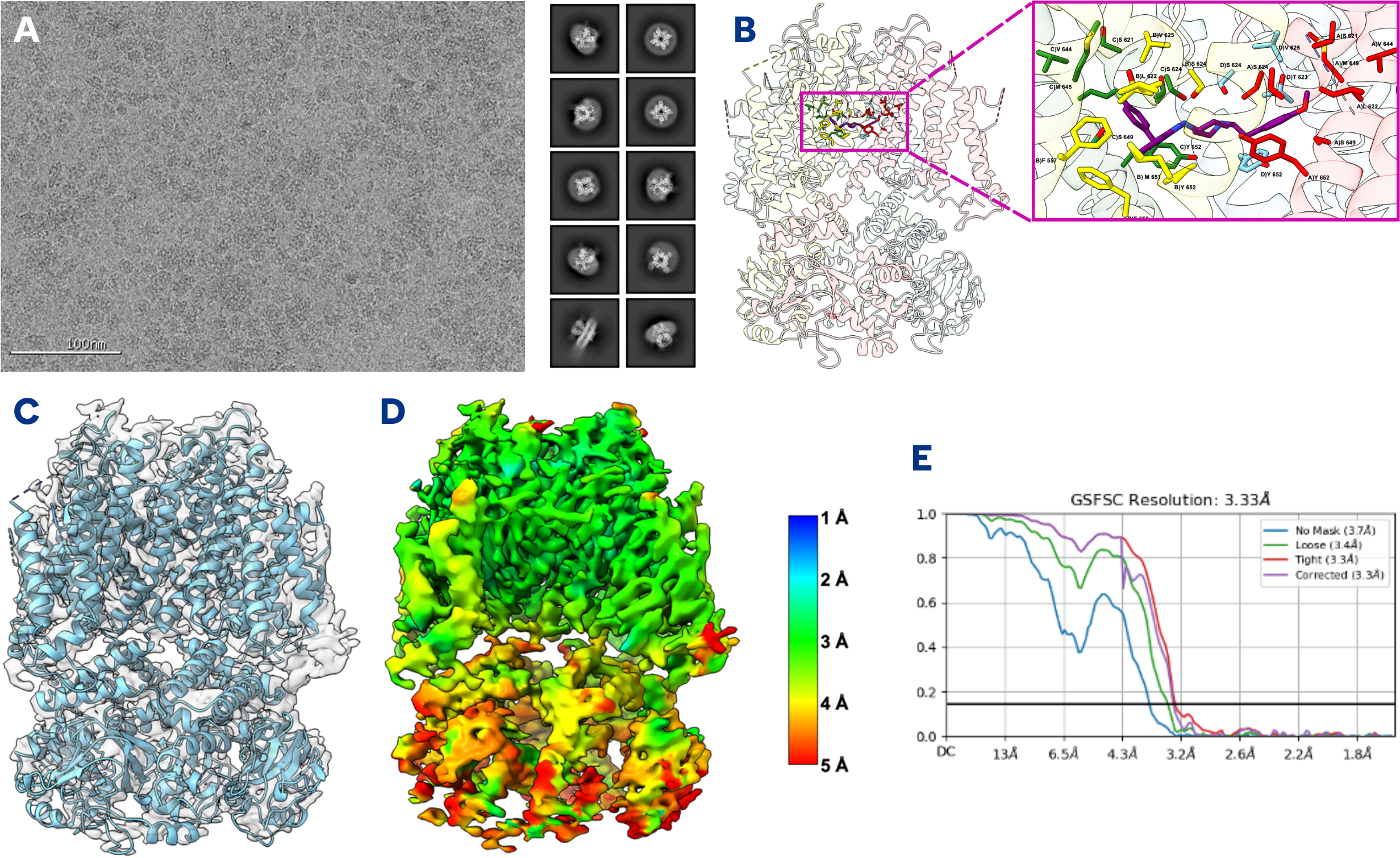
Accurate, Reliable Off Target hERG Structure Determination
The structure of the hERG channel is of great importance in:
- Drug Safety Assessment
- Drug Design and Optimization
- Mechanistic Insights
- Regulatory Guidelines
Analyze hERG-Ligand Complexes with Cryo-EM
Using high resolution cryo-EM imaging, NIS will provide a 3D structure reconstruction of detergent solubilized hERG in complex with your ligand. This includes:
- Vitrification
- Grid screening
- High resolution data collection
- Final refined 3D reconstruction to a resolution sufficient to enable visualization of the ligand

Target Classes Solved at NIS
Analyze hERG-Ligand Complexes with Cryo-EM
hERG Structure Determination in Drug Design and Optimization
hERG (human Ether-à-go-go-Related Gene) encodes a protein that is critical in the cardiac action potential, specifically forming the ion channel responsible for the rapid delayed rectifier potassium current. This current is crucial for the repolarization phase of the cardiac action potential, which helps in setting the rhythm and rate of heartbeats. The significance of hERG in drug development is due to its physiological role, but also to the fact that it represents an unwanted target for many pharmaceuticals, which can lead to unintended and potentially dangerous side effects.
- Drug Safety Assessment: The hERG channel has gained significant attention in the pharmaceutical industry because many drugs have been found to interact with it. Certain medications bind to the hERG channel and can block its activity, leading to a delay in repolarization. This delay, known as QT prolongation, can result in a life-threatening condition called Torsades de Pointes (TdP), which is a form of irregular heartbeat. Drugs that are found to inhibit hERG channels are subject to strict regulatory scrutiny and often require black box warning or may even be withdrawn from the market.
- Drug Design and Optimization: Understanding the structural basis of the drug:hERG interaction may be crucial for addressing the potential safety risks of new drugs and reducing or eliminating potentially harmful cardiac side effects. By analyzing the channel's three-dimensional structure, scientists can identify potential binding sites for drug molecules. Rational drug design can help minimize the risk of unwanted interactions with the hERG channel and reduce the potential for adverse cardiac effects.
- Mechanistic Insights: Understanding the structure of the hERG channel provides valuable insights into its functioning and the underlying mechanisms of cardiac electrophysiology. The channel's structure can reveal how it interacts with other molecules, including drugs and signaling proteins. This knowledge can lead to a better understanding of various heart-related disorders and the development of novel therapeutic strategies.
- Regulatory Guidelines: Regulatory authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), have established guidelines regarding the assessment of hERG-related cardiac risks for new drug candidates. These guidelines often require experimental data on a drug's interaction with the hERG channel. Therefore, having a comprehensive understanding of the hERG channel's structure and its relationship with drug binding is essential for complying with these regulatory requirements.
The structure of the hERG channel is of great importance in drug design and optimization, providing mechanistic insights into cardiac electrophysiology, and adhering to regulatory guidelines. Understanding the structure of hERG:drug complexes helps in minimizing the risk of cardiac side effects associated with drug interactions and aids in the development of safer and more effective medications.
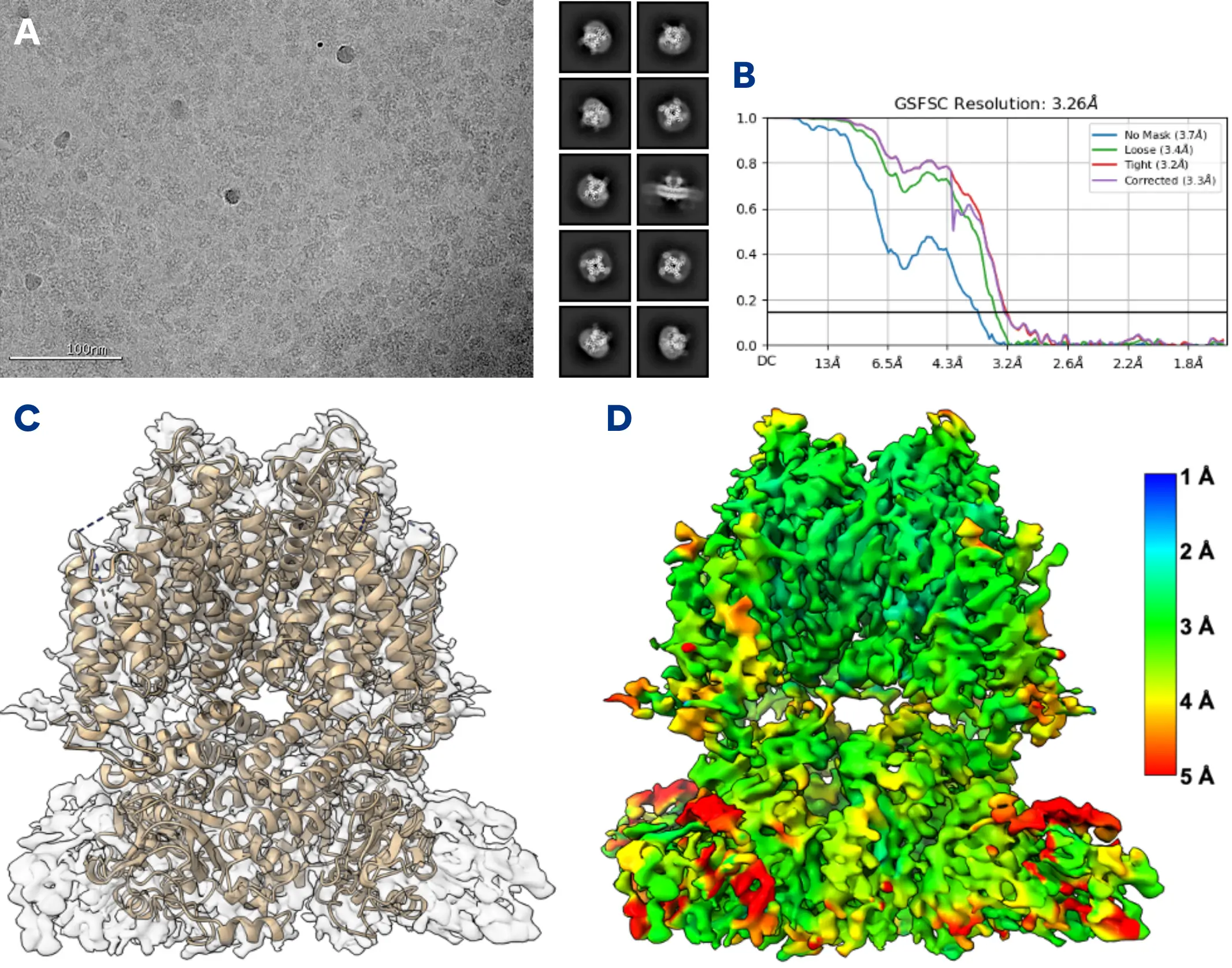
Using high resolution cryo-EM imaging, NIS will provide a 3D structure reconstruction of detergent solubilized hERG in complex with your ligand. This includes vitrification, grid screening, high resolution data collection, and a final refined 3D reconstruction to a resolution sufficient to enable visualization of the ligand. Model building can be added.